The dual incision surgical approach for total femur resection and endoprosthesis reconstruction in patients, with femur bone sarcoma: A case series
Received: 15-Sep-2022, Manuscript No. jotsrr-22- 77413; Editor assigned: 17-Sep-2022, Pre QC No. jotsrr-22- 77413 (PQ); Accepted Date: Oct 12, 2022 ; Reviewed: 01-Oct-2022 QC No. jotsrr-22- 77413 (Q); Revised: 07-Oct-2022, Manuscript No. jotsrr-22- 77413 (R); Published: 14-Oct-2022, DOI: 10.37532/1897- 2276.2022.17(10).73
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
Introduction: Limb preservation following the excision of tumors that involve the entire femur provides a therapeutic hurdle. Bone sarcomas involving almost the whole femur necessitate radical excision which encompasses removal of the entire femur bone, along with significant resection of the muscles involved in hip and knee function. Traditionally, surgical treatment of large femoral tumors required amputation via hip disarticulation, however, newer methods of preserving the limb, namely, Total Femur Replacement (TFR), have emerged. In comparison to amputation, limb preservation surgery is more cost-effective and better tolerated by patients. In the long run, it is associated with much lower oxygen use and energy requirements per meter walked.
Keywords
Distal radius fracture, intraarticular, augmented external fixator, volar plate fixation, mayo wrist score
Introduction
Limb preservation following the excision of tumors that involve the entire femur provides a therapeutic hurdle. Bone sarcomas involving almost the whole femur necessitate radical excision which encompasses removal of the entire femur bone, along with significant resection of the muscles involved in hip and knee function. Traditionally, surgical treatment of large femoral tumors required amputation via hip disarticulation, however, newer methods of preserving the limb, namely, Total Femur Replacement (TFR), have emerged. In comparison to amputation, limb preservation surgery is more cost-effective and better tolerated by patients. In the long run, it is associated with much lower oxygen use and energy requirements per meter walked [1-3]. TFR, specifically, is associated with better functional capacity and ambulation compared to hip disarticulation [4].
The use of total femur replacement is commonly reserved for malignancies that cover the majority of the entire femur. In these instances, an insufficient amount of bone remains to provide inadequate anchorage for modular implants or a custom-made joint-sparing endoprosthesis. The literature also establishes the use of TFR for extensive bone loss as a result of recurrent orthopedic fixation [5-7]. However, emphasis must be made on performing a safe, and structured anatomic approach, preventing unnecessary devascularization. This should provide satisfactory exposure to the whole bone with minimal soft-tissue damage [8-12].
Traditionally, TFR procedures utilized a single incision that extends a few centimeters proximal to the greater trochanter, curves anteromedially over the anterior thigh, and extends medially across the knee joint to the tibial tuberosity [8]. The literature does not address the implication of this extensive incision on wound healing and functionality. This technique also involves dissection through the thigh musculature and ligation of the healthy perforating vessels depriving the skin and subcutaneous tissue of this blood supply
Evidence demonstrates the occurrence of wound complications at a rate between 25%-61% and infection in 25% following the use of an extensible single incision method. The majority of patients with complications required readmission and excision of the necrotic region followed by delayed primary closure [9-11].
We have adopted a less invasive dual incision approach in an effort of reducing surgical trauma and minimizing the magnitude of sacrificed muscular tissue. The purpose of this study is to compare the outcomes of patients who have undergone the traditional TFR surgical approach vs the dual incision technique.
Material and Method
At the King Hussein Cancer Center (KHCC), a total of 9 patients underwent TFR from 2008 to 2020. Seven patients underwent primary resection of the bone tumour involving approximately the entirety of the femur which mandated reconstruction with an endoprosthesis. One patient was found to have extensive bone loss due to failed previous bone fixation. Another patient showed evidence of recurrent osteosarcoma following a previous distal femur replacement. This case necessitated subsequent resection of the remaining femur and conversion into TFR.
We retrospectively collected data on hospital stays and postoperative complications. Post-operative functionality of the limb was measured using the MSTS-ISOLS scoring system modified by Enneking et al. The score consists of 6 domains pain, function, emotional acceptance, use of supports, walking ability, and gait measured on a 5-point scale. In the first five cases, as Table 1. demonstrates, the single extensile surgical approach, previously described in literature, was used [6,8]. In the last four cases, the dual incision approach was established and applied by the senior author (AS). All of the TFR procedures were performed by the same surgeon.
Table 1. Demographic data of patients that had TFR in the KHCC
| Patient Number | Age | Diagnosis | Approach | Complication | Recurrence | Status | distant metastases | Margin | MSTS score |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 20 | Osteosarcoma | Single-incision technique | wound necrosis | yes | Dead | No | Negative | 0.73 |
| 2 | 50 | Osteosarcoma | Single-incision technique | none | no | Alive | No | Negative | 0.76 |
| 3 | 10 | Osteosarcoma | Single-incision technique | none | yes | Dead | Yes | Negative | 0.8 |
| 4 | 44 | Osteolysis | Single-incision technique | wound necrosis and deep infection | no | Alive | No | Negative | 0.67 |
| 5 | 15 | Ewing sarcoma | Single-incision technique | wound necrosis and deep infection | no | Alive | No | Negative | 0.56 |
| 6 | 5 | Ewing sarcoma | Dual incision technique | none | no | Dead | Yes | Negative | 0.87 |
| 7 | 7 | Osteosarcoma | Dual incision technique | none | no | Alive | No | Negative | 0.76 |
| 8 | 17 | Ewing sarcoma | Dual incision technique | none | no | Alive | No | Negative | 0.83 |
| 9 | 15 | Osteosarcoma | Dual incision technique | none | no | Alive | Yes | Negative | 0.83 |
SINGLE EXTENSILE SURGICAL APPROACH
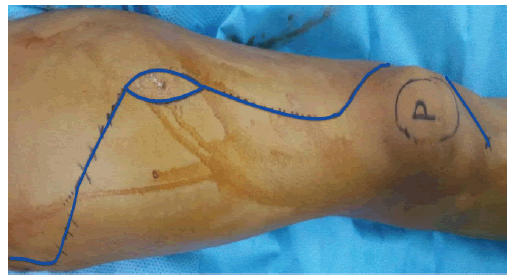
The patient was positioned in a semisupine with side support. The incision site was initiated 3cm-4 cm proximal to the greater trochanter, and curved anteromedial toward the midthigh. The incision was then made to continue medially to the knee and curve back to the midline to reach the tibial tuberosity Figure 1
This approach deprives a long segment of skin of its underlying muscular attachment, ultimately hindering the integrity of the anterior midthigh muscles
DUAL INCISION SURGICAL APPROACH
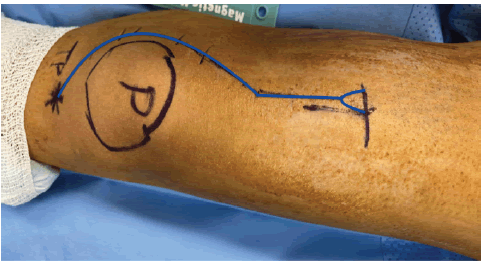
The distal incision (Figure 2) is constructed utilizing a standard anterior popliteal approach of the distal femur [12]. The length of the incision is determined by the extent of the extraosseous soft tissue component to be exposed in an effort to achieve complete dissection. Complete dissection encompasses all non-involved muscle tissues, femoral vessels, and sciatic nerve away from the tumor to perform knee arthrotomy and release of the medial and lateral gastrocnemius muscle from the distal femur. This is followed by the exploration and isolation of the popliteal vessels posteriorly away from the tumor.
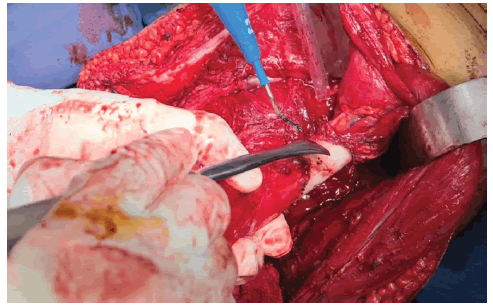
Then, the muscle attachment of the distal femur is released. Posteriorly, a knee capsulotomy will be performed with dissection of the Anterior and Posterior Cruciate Ligaments (ACL and PCL) in effort to completely dislocate the femur Figure 3.
The proximal incision uses the same technique starting 2 cm above the greater trochanter, then curving the incision slightly anterior as in Figure 4.
The incision is then advanced distally over the lateral shaft of the femur,to identify the insertion of the gluteus maximus muscle, tag it and release it. It is necessary to also identify, label, and detach the insertion of the gluteus medius, minimus at the greater trochanter, and the origin of vastus lateralis. We also isolate the external rotators from proximal to distal: piriformis, superior gemiulls, obturator internus, inferior gemillus, and quadratus femoris.
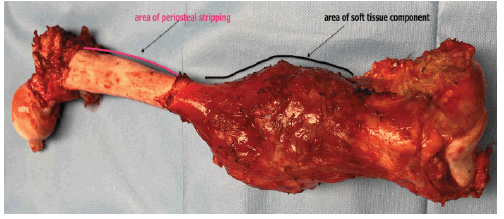
Subsequently, the release of muscular attachment at the proximal femur from the anteromedial side, including the pectinus and iliopsoas muscles is possible. In cases in which the tumour presents entirely intraosseous, the periosteal elevator may be used to strip the periosteum and muscles from their bony attachment Figure 5.
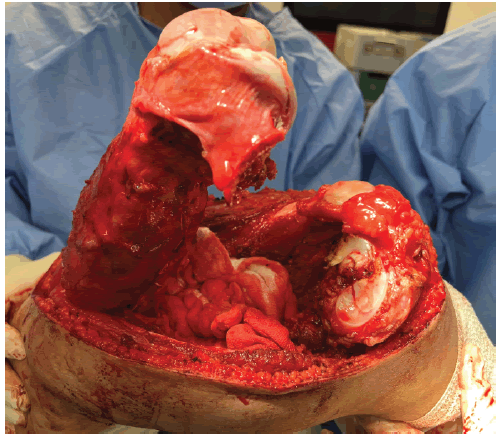
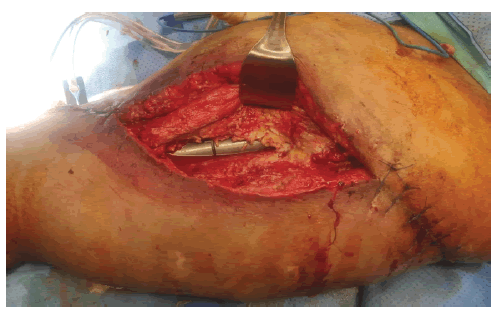
Afterward, the surgeon may proceed with the detachment distally without jeopardizing the thick musculofasciocutaneous flap between the 2 skin incisions. The entire femur may then be pulled from the distal wound to the exterior of the body Figure 6.
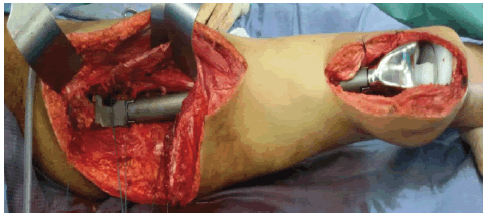
Following complete resection, the proximal tibial articular surface is prepared to receive the passive tibial component, and the modular total femur endoprosthesis is inserted Figure 7.
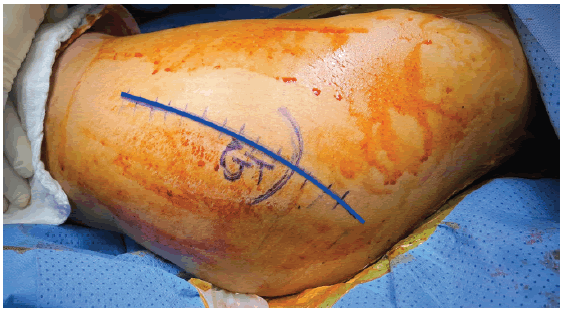
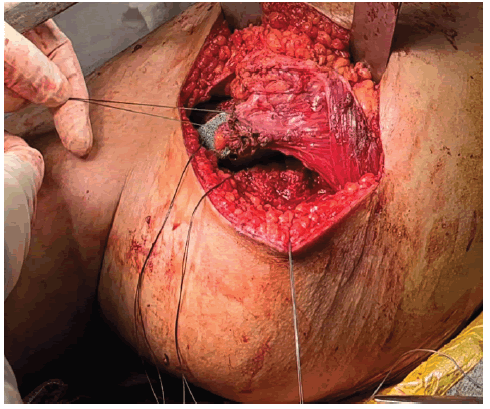
Dacron tape is used to reattach the abductor tendons, vastus lateralis, and gluteus maximus to the implant Figure 8.
Finally, drains may be inserted, closure in layers performed, knee and hip stabilizers are applied. In terms of post-operative rehabilitation, we strictly follow our previously published protocols [13].
IMPLANTABLE FEMUR
For total femoral replacement in these patients, we utilized the Global Modular Replacement System (GMRS) by Stryker as shown in Figure 9.
This prosthetic device is reserved for patients undergoing Total Femur Replacement (TFR).
Result
Data collected from osteosarcoma patients at the KHCC is depicted in Table 1. None of the 9 patients (0%) showed evidence of dislocation nor complications that necessitate implant revision.
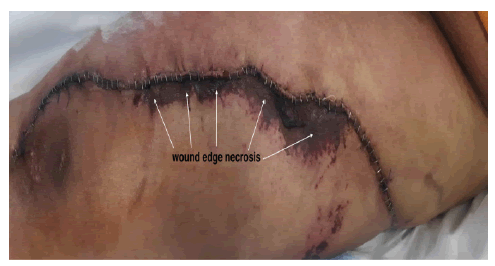
The first 5 patients which received the previously published extensile single incision approach. These patients all showed evidence of negative resection margins. Postoperative hospital stay was 24 hours in the surgical ICU for observation followed by transfer to the surgical ward. All patients received blood transfusion perioperatively consisting of 4 units-8 units of packed red blood cells and were discharged on postoperative days [6-10]. Three of the 5 patients undergoing a single incision were readmitted due to the development of wound edges necrosis as seen in Figure 10
Similarly, two cases resulted in deep periprosthetic infection as well Figure 11 requiring subsequent hospitalization, an average of 7 debridement sessions [6-10], and multiple bedsides and operative vacuum dressings. Intravenous systemic antibiotics were administered based on the yielded microorganisms in the intraoperative culture. The average added hospital stay needed for the management of wound complications was 36 days (ranging from 18-54). The three patients retained their limb and endoprosthesis, however, 2 patients had a chronic infection and draining sinus. The average modified MSTS/ ISOLS functional score for those 5 patients was 70%.
The latter four patients illustrated in Table 1 underwent the dual incision approach. This subgroup of patients also remained in the surgical ICU postoperatively for 24 hours before being transferred to the surgical floor. All patients within this group received blood transfusions perioperatively ranging from 2 units to 5 units of packed red blood cells. On the 6th or 8th postoperative day, patients were discharged and did not necessitate readmission. The incisions in the group healed adequately allowing for the removal of the stitches three weeks after surgery. The average modified MSTS /ISOLS functional score for the 4 patients was 82%.
Discussion
To our knowledge, this is the first case series to compare the use of dual incisions in TFR for bone sarcoma patients to patients treated using the previously reported extensile single incision. The dual incision technique aims to minimize surgical trauma to the skin, subcutaneous tissues, and fat by preserving an intact area of soft tissue between the proximal and distal wounds. Ultimately this may lead to improvement of the wound healing process yielding a decrease in the occurrence of wound edges and muscular necrosis. In addition, by decreasing the magnitude of trauma to the musculature of the anterior thigh, this, in theory, may retain a larger amount of functional muscle tissue.
As part of the quadriceps muscle group, the Vastus Lateralis (VL) contracts during the termination of the swing phase of gait to prepare the knee for weight-bearing, the muscle group as a whole is responsible for absorbing the vast majority of the force generated by the heel strike. The muscle group continues to contract through the early portion of the stance phase as part of the loading response. The VL is the strongest member of the quadriceps muscle group, and thus it is estimated to contribute approximately 40% of the overall strength of the quadriceps muscle group, with Rectus Femoris (RF) and Vastus Intermidus (VI) accounting for 35% and the Vastus Medialis (VM) totaling the last 25% [14]. As a result, this may improve postoperative function following rehabilitation.
Therefore, this operation may improve postoperative function following rehabilitation. We noted a tendency toward the improvement of functionality in patients undergoing the dual incision technique compared to that of the single incision. Moreover, we noted a decreased predilection in the occurrence of wound complications, infection, readmission, and subsequent debridement. We found a higher propensity of wound complications which occurred in 3 out of 5 patients who underwent a single incision for TFR which was supported by previous reports [9-11,13]. Thus, we recommend the adjustment of surgical incisions to accommodate two smaller incisions leaving intact tissue in between. Subperiosteal dissection of muscles attached to the involved bone with high-grade sarcoma may be safely performed in areas with no cortical breakage and absent soft tissue components of the tumour. This study has some limitations. First, this study is retrospective. However, the surgical indications were consistent throughout the timeframe included in this study. Second, the final cohorts are relatively small.
Conclusion
The dual incision approach is proved to be oncologically safe, as the tumor may be resected completely with negative margins. Moreover, these patients show no added operative time or blood loss. Our study proposes the utilization of a new dual incision approach for TFR in patients with femoral sarcoma. Due to the rarity of Total Femur Resection (TFR), in orthopedic practice, it is not possible to conduct a case-control comparative study to assess the advantages of dual incision techniques over the extensile single incision. Therefore, recommend larger multicenter collaborative studies to provide concrete evidence of the proposed superiority of a dual incision over the traditional single extensile approach.
References
- Grimer RJ, Carter SR, Pynsent PB. The cost-effectiveness of limb salvage for bone tumours. J Bone Joint Surg Br 1997;79-B(4): 558-61. [Google Scholar][CrossRef]
- Otis JC, Lane JM, Kroll MA. Energy cost during gait in osteosarcoma patients after resection and knee replacement and after above-knee amputation. J Bone Joint Surg Am 1985;67-A(4):606-10. [Google Scholar][CrossRef]
- S Kalra, A Abudu, H Murata R et al. Total femur replacement: Primary procedure for treatment of malignant tumours of the femur. 36(4), 383. [Google Scholar][CrossRef]
- Adel Refaat Ahmed (2010). Total femur replacement. 130(2), 171-176. [Google Scholar] [CrossRef]
- Agrawal A, Agrawal V, Yadav S, et al. A Case Report of Dual Incision Technique for Total Femur Arthroplasty as a Salvage Procedure in Infected Non- Unions. J Orthop Case Rep. 2017 Jul-Aug;7(4):44-47. [Google Scholar][CrossRef]
- Bickels, Jacob Meller, Isaac Henshaw, et al. (2000). Reconstruction of Hip Stability After Proximal and Total Femur Resections. Clin Orthop Relat Res. 375(), 218;230. [Google Scholar][CrossRef]
- Gosheger, Georg Gebert, Carsten Ahrens, et al. (2006). Endoprosthetic Reconstruction in 250 Patients with Sarcoma. Clin Orthop Relat Res. 450(), 164-171. [Google Scholar][CrossRef]
- Malawer M, M & Sugarbaker P H. (2001). Musculoskeletal cancer surgery treatment of Sarcomas and allied diseases. Kluwer Academic. [Google Scholar][CrossRef]
- Toepfer Andreas, Harrasser Norbert, Petzschner Isabel et al. Short- to long-term follow-up of total femoral replacement in non-oncologic patients. BMC Musculoskeletal Disorders, 17(1), 498-.6 [Google Scholar][CrossRef]
- KL Pan, Chan WH, Shanmugam P et al. Large Volume Osteosarcomas of the Femur Treated with Total Femoral Replacement. Malaysian Orthopaedic Journal 8 (TFR): 5. [Google Scholar][CrossRef]
- Amanatullah, Derek F, Trousdale, et al. (2014). Non-Oncologic Total Femoral Arthroplasty: Retrospective Review. The Journal of Arthroplasty, 29(10), 2013;2015. [GoogleScholar][CrossRef]
- Chia-Chun Wu, Tamir Pritsch, Ahmad Shehadeh et al. (2008). The Anterior Popliteal Approach for Popliteal Exploration, Distal Femoral Resection, and Endoprosthetic Reconstruction. 23(2),254-262. [GoogleScholar][CrossRef]
- Shehadeh A, Dahleh M E, Salem A, et al.(2013). Standardization of rehabilitation after limb salvage surgery for sarcomas improves patients outcome. Hematolog Oncology and Stem Cell Therapy, 6(3-4), 105;111. [Google Scholar][CrossRef]
- Farahmand F, Sejiavongse W, Amis AA. Quantitative study of the quadriceps muscles and trochlear groove geometry related to instability of the patellofemoral joint. Journal of Orthopaedic Research. 1998 Jan;16(1):136-43. [Google Scholar][CrossRef]



 Journal of Orthopaedics Trauma Surgery and Related Research a publication of Polish Society, is a peer-reviewed online journal with quaterly print on demand compilation of issues published.
Journal of Orthopaedics Trauma Surgery and Related Research a publication of Polish Society, is a peer-reviewed online journal with quaterly print on demand compilation of issues published.